Reimbursement
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.
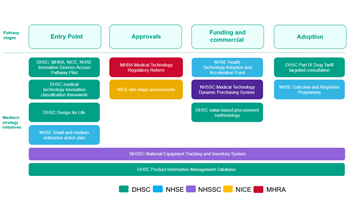
UK Medtech Strategy Sets Out Schedule Of Milestones To FY 2026
Fourteen months on from the release of its inaugural medtech strategy, the UK MedTech Directorate has laid firm foundations and reports progress on initiatives aimed at improving technology adoption. A schedule of ambitious future timelines has also been published.

It’s Been Over 1100 Days Since MCIT, Still No TCET In Sight
AdvaMed’s new online clock documents the time since CMS promised to replace MCIT, a program guaranteeing coverage for FDA-cleared breakthrough devices.

Ekso's Exoskeleton Receives Medicare Coverage, Paves Way To Faster Rehabilitation
Ekso Bionics’ exoskeleton received a final payment determination from the Centers for Medicare and Medicaid Services, paving the way to faster rehabilitation for more patients.

News We’re Watching: Lung Preservation System Goes National, De Novo For MMI, Brazil’s ANVISA To Recognize Overseas Approvals
This week, Paragonix announced that its BAROguard lung preservation system is now available throughout the US; Brazil planned to leverage some foreign device approvals; Medical Microinstruments’ Symani Surgical System won de novo clearance; and the FDA updated its safety warnings for Essure and certain plastic syringes.

FDA Asked To Withdraw Approval Of Test Evaluating Opioid Addiction Risk
Dozens of researchers signed a letter condemning the AvertD test from SOLVD, which looks for genes that may contribute to a propensity toward opioid addiction. But the FDA and the manufacturer both say the test is effective.
Abbott Wins FDA Approval For TriClip Repair System, Compete Against Edwards
Abbott wins FDA approval for TriClip regurgitation therapy, which will compete against Edwards Lifesciences’ recently approved Evoque transcatheter tricuspid valve replacement device. Two analysts expect Tricuspid will be eligible for a new technology add-on payment, which will be positive for Abbott.

Collecting Sensitive Data With AI Chatbots
Medtech Insight spoke to QliqSOFT executive Bobbi Weber about the advantages of collecting patient data though chatbots, and how the technology can make conversations at the doctor’s office a little more manageable.

FDA Leader Looks To International Effort On Pediatric Device Development
Collaboration with Japan and Europe could help ease the development path for pediatric devices, cardiovascular device office director Bram Zuckerman said at a recent conference. Zuckerman also spoke on other cardiac device priorities at the FDA.

‘Indomitable Spirit’ Spurs Incontinence Treatment Firm Axena
Axena Health recently secured a HCPCS code for its female incontinence device Leva. For CEO Eileen Maus, this is just the latest in a string of successes across the women’s health, or femtech, sector.

News We’re Watching: New Cybersecurity Standard; CMS Prodded On TCET; Olympus Scopes Recalled
This week, NIST published a new version of its international cybersecurity standard; SeaStar Medical’s kidney device got an HDE; and the FDA announced safety issues for DT MedTech LLC, operating room tables, and GE incubators.

DOJ: False Claims Settlements Neared $2.7Bn In 2023
The government collected $2.68bn from settled False Claims Act cases last year, the US Department of Justice said. Another 12 device-related cases were announced in February alone.

NICE Recommends Endoscopic Sleeve Gastroplasty To Treat Obesity
NICE has recommended endoscopic sleeve gastroplasty for the treatment of obesity in adults.
SVB’s Milo Bissin Predicts ‘Reckoning In 2024’ For Health Care Companies With Sky-High Valuations
VC funding will remain challenging for many in the health care industry, Silicon Valley Bank’s market manager Milo Bissin told attendees at the recent DHIS West conference. Companies focusing on promising areas such as AI-powered technologies, women’s health and value-based care likely have the best chances to draw investors’ attention.

UK Offers £10M Funding To Eight Medtech Innovators
The Innovative Device Access Pathway pilot has launched in the UK with eight new promising medical technologies. The cohort is overwhelmingly diagnostic-focused, with innovative testing for Alzheimer’s, stroke, and liver cancer, amongst others.
$2Bn Hike In Catheter Billings Could Point To Fraud
The US government is said to be investigating a sudden spike in Medicare billings for urinary catheters, which jumped from $153m in 2021 to $2.1bn in 2023.

Progress Made With HTA Regulation Implementation Plan
Implementing acts for Joint Clinical Assessments and exchange of information with the EMA were on the agenda of this month’s Member States Coordination Group on HTA meeting. The discussion marks considerable progress towards the application deadline in January 2025.
You must sign in to use this functionality
Authentication.SignIn.HeadSignInHeader
Email Article
All set! This article has been sent to my@email.address.
All fields are required. For multiple recipients, separate email addresses with a semicolon.
Please Note: Only individuals with an active subscription will be able to access the full article. All other readers will be directed to the abstract and would need to subscribe.