COVID-19 Watch
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

At-Home Health Testing Demand Is High Post-Pandemic, But So Are Barriers To Development And Use
At the recent Precision Med-Tri Con conference, laboratory experts traded views on the expansion of at-home testing for disease diagnosis and personalized health insights. While strong consumer demand spells opportunity, there are significant concerns about the accuracy and reliability of home-testing platforms, misuse, accessibility, and lack of health literacy.
Ultrahuman Expands Wearable Medtech Production Into US After $35M Funding Round
Firm operating in London, India and United Arab Emirates says its “Ultra Factory” will open in Indiana within the next six months with end-to-end production based on its operational facility in India.

What Devices Are Essential During A National Emergency? FDA Panel Makes A List
Members of the US FDA’s General Hospital and Personal Use Devices Panel of the Medical Devices Advisory Committee met on 6 February to discuss what medical devices are critical to public health during a national emergency and help the FDA create a list of those devices.

Califf Touches On A Range Of Issues And Priorities During Fireside Chat
During a recent webinar hosted by the Alliance for a Stronger FDA, commissioner Robert Califf discussed the agency’s 2024 budget, its ongoing reorganization, AI, LDTs, and the continuing problem of misinformation.

‘Banner Year’ For FDA’s Device Center Includes Record Number Of Novel Authorizations
A new annual report argues that last year was both transformative and historic for the US FDA’s Center for Devices and Radiological Health. The center authorized the highest number of novel medical devices in history while turning the corner on the pandemic.

New FDA List Shows 18 COVID-19 Tests With Traditional Authorization
Manufacturers including BioFire, Cepheid and Roche have already secured multiple 510(k) or de novo clearances from the US FDA. However, more than 400 COVID-19 diagnostics remain on the market under EUAs.

Former Diagnostics CEO Pleads Guilty In Securities Fraud Case
Keith Berman claimed to investors that his company, Decision Diagnostics, was on the verge of releasing a groundbreaking COVID-19 test. The problem? It wasn’t true.

FDA Issues Final Guidance On Device Shortage Reporting, Adds Device List
The US FDA has published final guidance requiring firms to notify the agency about significant manufacturing interruptions of medical devices during a public health emergency, as well as a supplementary draft guidance that includes a list of specific devices that manufacturers must report to the agency when they are in short supply.

News We’re Watching: Expiring Guidance Documents; Multi-Cancer Diagnostic Meeting Set; 510(k)s For BD And Smileyscope
This week, the US FDA announced that several pandemic-era guidance documents are no longer in effect; an FDA advisory committee meeting on multi-cancer detection (MCD) diagnostics was scheduled; and both a needle-free blood collection device and the first VR treatment for pain and anxiety landed FDA clearances.
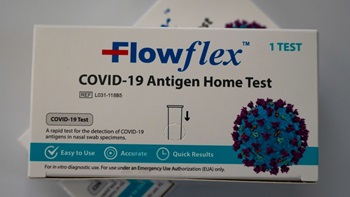
Flowflex Becomes First At-Home COVID-19 Antigen Test Cleared Through 510(k)
The test is among the first of a wave of products initially authorized for emergency use by the US FDA that are now pursuing traditional approval pathways to stay on the market.

Amazon To Offer Prime Members Low-Cost Access To One Medical Primary Care
Amazon announced it will offer Prime members a One Medical subscription for $9 a month, or $99 a year in a move to “make it easier for people to get the health care they need.”
Prepping For Flu Will Boost Pandemic Readiness, Baldwin Says
Legislation introduced in the US Senate by Wisconsin’s Tammy Baldwin would beef up the nation’s flu preparedness by investing in new technology for vaccine and diagnostic development as well as treatments.

News We’re Watching: AGs Push For Pulse Ox Warnings. VP Harris Announces AI Initiatives; FDA Looks At Digital Health And Diabetes
This week, 25 state attorneys general pressed FDA Commissioner Robert Califf to improve pulse oximeter safety warnings; MDIC extended the deadline on an industry cybersecurity benchmark study; and more guilty pleas and settlements were entered in Medicare fraud cases.

Report: Problems At CDC Delayed First COVID-19 Test
Lack of laboratory quality management procedures and resource shortages were behind the CDC’s troubles in developing a COVID-19 diagnostic in 2020, an HHS OIG audit found.

The FTC Complaint Is Fully Visible. The COVID-19 'Mask' Is Not.
New York business partners continued false advertising for “The 1 Virus Buster Card,” to be worn around the neck or clipped onto clothing, on their website and social media platforms after receiving a warning from the US FTC in July 2020.

Trade Commission Finds Mixed Effects From Patents In COVID-19 Fight
A new report from the US International Trade Commission found that patents had a mixed effect on access to diagnostics and therapeutics during the COVID-19 pandemic. Trade group AdvaMed says the findings demonstrate that patent waiver programs aren’t needed for COVID-19 diagnostics.
You must sign in to use this functionality
Authentication.SignIn.HeadSignInHeader
Email Article
All set! This article has been sent to my@email.address.
All fields are required. For multiple recipients, separate email addresses with a semicolon.
Please Note: Only individuals with an active subscription will be able to access the full article. All other readers will be directed to the abstract and would need to subscribe.